Calls Now Open
2027 Scholar-Innovator and ADDF-Harrington
May 05, 2016
For the past decade, venture philanthropists have been working to propel promising therapies and vaccines into the clinic, with some success.
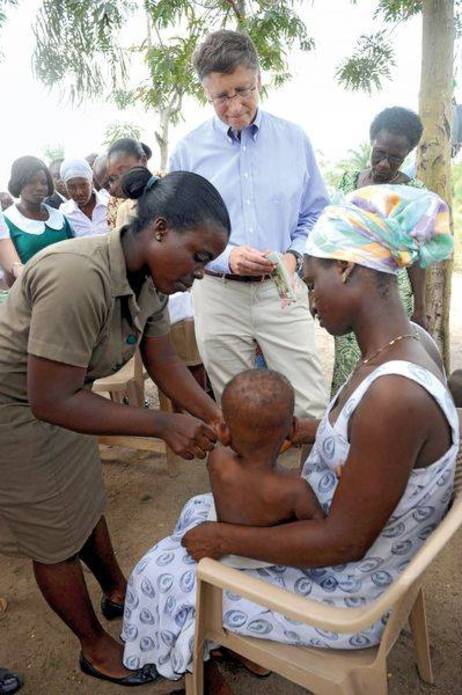
Venture philanthropist Bill Gates looks on as a health worker vaccinates a child in Ghana
In December 2015, Facebook founder Mark Zuckerberg and his wife Priscilla Chan made a stunning announcement. In a letter to their newborn daughter, the couple pledged to give away 99% of their Facebook stock — about US$45 billion. Part of that money, they said, will go towards finding therapies for five global killers: heart disease, cancer, stroke and neurodegenerative and infectious diseases. “Curing disease will take time,” Zuckerberg wrote. “Over short periods of five or ten years, it may not seem like we're making much of a difference. But over the long term, seeds planted now will grow, and one day, you or your children will see what we can only imagine: a world without suffering from disease.”
But Zuckerberg and Chan aren't inclined to simply write a cheque. They are part of a cadre of philanthropists taking a more hands-on approach. These venture philanthropists hope to leverage their business savvy to shepherd new therapies to market — fast. “They want to roll up their sleeves and understand how their dollars are being used to address unmet needs, to overcome research roadblocks and to take advantage of promising new discoveries,” says Melissa Stevens, executive director of the Center for Strategic Philanthropy at the Milken Institute in Washington DC. Max Wallace, chief executive of Accelerate Brain Cancer Cure, or ABC2, in Washington DC, puts it more bluntly: “These type of new rich don't want to look like fools. They don't want their money to be wasted.”
Microsoft co-founder Bill Gates has become the poster child for venture philanthropy. Since 2000, the Bill & Melinda Gates Foundation has poured more than $20 billion into global health. But many of the basic tenets of the model arose more than a century ago. “When philanthropy was developing in America there was this idea that foundations had this great capacity, because they weren't the government, to solve social issues and be really innovative and take big risks,” says Alexandra Graddy-Reed, who studies non-profit organizations and their policies at the University of Southern California in Los Angeles. “Carnegie and Rockefeller had many of these principles when they were giving a hundred years ago.”
In the past decade venture philanthropy has experienced a resurgence, with many foundations focused on new therapies. But the attributes that make this type of funding so effective can also stir up controversy or raise ethical questions. Philanthropic foundations are not accountable to the public, and some critics question whether wealthy benefactors have too much sway in medicine.
Taking Risks
The US National Institutes of Health invests about $32 billion in biomedical research and development each year, much of which goes towards basic research. If a new therapy looks promising, “the expectation was that for-profit venture capitalist companies would come in and help those academic researchers spin out successful cures or drugs,” says Graddy-Reed, and then big pharmaceutical companies would take over. But in recent years, the system has broken down. According to a report by US trade association the Biotechnology Industry Organization, venture funding of private drug-development companies peaked in 2007 at $5 billion. Then the financial crisis hit and funding fell by nearly half, to $2.8 billion in 2010. Investments have begun to recover, but they were still below pre-crisis levels in 2014. Although 2015 was a banner year for drug and biotech companies seeking venture capital, industry experts point out that early-stage research is still underfunded. Increasingly, venture philanthropy is stepping in to fill the gap. “It has emerged as the industry's new high-risk capital,” Stevens says.
In many ways, philanthropic funding is well suited to the task. “We can take risks that neither governments nor the private sector can afford to take. We don't have the same pressures for monetary return,” says Penny Heaton, director of vaccine development at the Bill & Melinda Gates Foundation. “Our metrics are all about saving lives.” This appetite for risk allows foundations to fund early-stage drug development, and even support unorthodox approaches. “They can really feed exploration in scientific areas where others might not be willing to go because it's so new or so innovative,” Stevens says. For example, the Stanley Medical Research Institute — founded in 1989 by Ted and Vada Stanley, whose son was diagnosed with bipolar disorder — supports research that investigates infectious agents such as the parasite Toxoplasma gondii as possible cause of schizophrenia. “When we started our research on infectious agents 25 years ago, it would have been impossible to get government funding,” says psychiatrist E. Fuller Torrey, associate director for research at the institute.
But moving drugs through the pipeline takes more than funding. “If money were the solution, I think this problem would have been tackled long ago,” says Jonathan Stamler, director of the Harrington Discovery Institute in Cleveland, Ohio.
Stamler was a cardiovascular researcher at Duke University in Durham, North Carolina, when the financial crisis hit, and he watched with alarm as funding for drug development dried up. “It became increasingly difficult to find a way to move discovery forward,” he says. So Stamler came up with an approach to fund early-stage innovators — an organization that would behave as both a non-profit institute and a for-profit company.
After relocating to University Hospitals Case Medical Center in Cleveland in 2010, Stamler paired up with Baiju Shah — who had extensive experience of launching biomedical companies — and together they took Stamler's idea to local philanthropist Ronald Harrington. Harrington and his family had already donated money to support cardiovascular research after he had a quadruple bypass in 2000. But Stamler and Shah pitched a way for the Harringtons to have an even greater impact on medicine: the non-profit institute would provide researchers with funding and much-needed industry expertise, and the for-profit accelerator would develop the most promising discoveries and hand them off to pharmaceutical firms to carry forward.
“They came at us four times,” Harrington recalls. Eventually, the family agreed, and the Harrington Project for Discovery and Development was born. The Harringtons donated $50 million to kick-start the non-profit arm, the Harrington Discovery Institute, and drummed up another $100 million in support from other donors. The Harringtons also invested an undisclosed, but much smaller, amount in the for-profit arm, BioMotiv. Harrington may have been sceptical at the outset, but he has since become an champion. “This opens up collaboration like no other model,” he says.
The project is just four years old, but already BioMotiv has brokered deals with several major pharmaceutical companies. Goutham Narla, a medical geneticist at Case Western Reserve University in Cleveland, thinks that his discoveries would have languished if he hadn't been selected as a 2012 Harrington Distinguished Scholar. “We just don't have the depth of pharmaceutical expertise in academia to do what I'd call true drug development,” he says. The Harrington Discovery Institute helped Narla to develop an anticancer therapy, and now his company, Dual Therapeutics, is part of BioMotiv. “We have weekly calls with people who have, collectively, 80-plus years of experience in pharma,” he says, and access to that experience is paying off. In January, BioMotiv announced that Dual Therapeutics would partner with drug giant Bristol-Myers Squibb. “The goal is to hopefully do clinical trials next year,” Narla says.
Many foundations have their roots in personal tragedy. As Stevens and her colleagues at the Center for Strategic Philanthropy like to say, “You don't go to medical philanthropy — medical philanthropy comes to you.” For the Case family, tragedy struck in 2001, when 43-year-old investment banker Daniel Case was diagnosed with an aggressive type of brain tumour called a glioblastoma. “2001 doesn't seem like that long ago. But in brain-cancer terms, it's kind of the dark ages,” Wallace says. Finding out that there were not any drugs available, Case enlisted the help of his brother Steve, the co-founder of digital media company AOL. Together with their families, the brothers founded ABC2.
Like Stamler, Wallace doesn't see money as the main barrier to drug development. ABC2, a 5-person foundation, has handed out only about $22 million in grants since 2001, and Wallace says that these days the foundation spends just $2–3 million a year. That may not be enough to fund clinical trials, but the money helps to bring people together. “Our role has often been to be a bio-yenta. Let's make some marriages,” he says. One particularly fruitful marriage began at the ABC2 2012 Annual Scientific Meeting in Sausalito, California. Wallace and his colleagues struck up a conversation with William Sellers, global head of oncology at Novartis Institutes for BioMedical Research, headquartered in Cambridge, Massachusetts. Novartis had been looking at combination therapies for cancer, but it wasn't developing any drugs for brain tumours. When Wallace asked why, Sellers explained that they needed tumour tissue.
Brain-tumour samples can be tricky to extract, but Wallace knew that neurosurgeons at the Henry Ford Hospital in Detroit, Michigan, had a reputation for having 'magic hands'. So Wallace asked Tom Mikkelsen, co-director of the Hermelin Brain Tumor Center at Henry Ford, to join the discussion. Within days, Mikkelsen had samples ready for Novartis. These allowed the company to generate mice with human brain tumours that they could use to screen Novartis's compound library for therapies. “We're really small, but we're trying to cast a big shadow,” Wallace says. “We've made $20 million worth of grants, but we have backed research that's led to 14 therapies being in the clinic.”
Cautionary tale
Although additional funding for biomedical research may seem like a winning proposition, the attributes that make philanthropic funding so powerful can also become stumbling blocks. Foundations can take more risks because they aren't accountable to the public. But this lack of accountability can also be a cause for concern. “We've given all these organizations, these individuals, a huge tax reprieve,” Graddy-Reed says. “But the public has no say in how the foundations spend their money.”
In some cases, philanthropic organizations are so large that they can drive the research agenda in a given area by themselves. The Gates Foundation, for example, has given away more than $36 billion since its inception. Around half of that has gone to global health, making the foundation the largest private supporter in that arena. “Their budget dwarfs the budget of small countries,” says Gregg Gonsalves, a researcher at Yale Law School in New Haven, Connecticut. In 2014, the Gates Foundation supplied $2.9 billion (or 8%) of the $35.9 billion that high-income countries provided to support global health. In some cases, the foundation's impact is even larger. The same year, the foundation gave 13.9% of the total funding for maternal, newborn and child health, and 12.6% of the total funding for tuberculosis.
“Because of their size, they have this huge ability to influence what it is we're trying to do as a society,” says Graddy-Reed. Whether that influence is a boon or a burden is a matter of debate. In a 2008 memo obtained by the New York Times, the then-director of the World Health Organization's malaria programme Arata Kochi, wrote that the foundation's tendency to push its favourite research “could have implicitly dangerous consequences on the policymaking process in world health”. Kochi is one the few outspoken critics of the foundation. In certain fields, nearly everybody has some involvement with the Gates Foundation, Gonsalves says, and “nobody is going to want to bite the hand that feeds them.”
Gonsalves acknowledges that the Gates Foundation has done a lot of admirable work, but he worries about the influence it could be having at a global level. The foundation is one of the largest funders of the World Health Organization. In 2014–15, it gave the organization $423 million — less than the United States, the agency's biggest donor, but more than the United Kingdom donated. That money is earmarked for particular projects. For example, nearly 70% of the Gates Foundation's 2014–15 contribution went to polio eradication. Ultimately, philanthropists have their own viewpoints and priorities, Gonsalves points out, and those drive the research agenda of their foundations.
Venture-philanthropy funding often comes with strings that can make commercialization more difficult, according to Kelly Sexton, director of the Office of Technology Transfer at North Carolina State University in Raleigh. Foundations might want royalties that are so high that the university is left empty handed. Or they may stipulate that the research be allowed to be licensed to multiple parties, which means that “when we go to find a licensee, we can't offer an exclusive license”, Sexton says. That can make it next to impossible to find companies willing to take up the technology.
When a foundation benefits financially from a drug that it helped to develop, the ethics can be murky. Since 2000, the Cystic Fibrosis Foundation has poured $150 million into Vertex Pharmaceuticals and another company that Vertex acquired to develop new drugs for the disease. In return, the foundation negotiated to keep some royalties. The investment paid off in 2012, when the US Food and Drug Administration approved Kalydeco (ivacaftor) — the first drug to treat the underlying cause of some forms of cystic fibrosis. “Funds from any royalties we receive are reinvested into further research and drug development and advance our mission to find a cure,” the president and chief executive at the time Robert Beall wrote in 2014, when the royalty rights were sold for $3.3 billion.
Some see the Kalydeco example as a success story of how non-profits can work with industry to bring much-needed drugs to market. But others have criticized the foundation for failing to negotiate a better price for the drug, which costs about $300,000 per person annually. Lisa Schwartz, a medical-communication researcher at Dartmouth Institute for Health, Policy and Clinical Practice in Lebanon, New Hampshire, notes that developing effective drugs is only part of the equation. “Isn't there some responsibility to provide access?” she asks.
Schwartz also says that having a stake in the sale of a drug intended for the patient group you're trying to serve creates a conflict of interest. “If you personally benefit every time that drug is prescribed, then the question is, will you fairly represent that drug?” she says. “Can you be the honest broker?”
The Cystic Fibrosis Foundation deal has prompted other organizations to ask whether they should adopt a similar model. “I am certain that after the announcement of the sale, every medical-research foundation in the US had a call with their board,” says Stevens. But despite the potential windfall, some have decided to forgo the profits. “They don't want to run the risk of being seen as a non-neutral party,” she says.
Zuckerberg and Chan's philanthropic plan is even more controversial. Rather than launching a foundation, the couple has set up a limited-liability company whose mission is “advancing human potential and promoting equality,” according to the couple. Unlike a foundation, a limited-liability company can freely invest in for-profit organizations without disclosing those investments, make political contributions and lobby governments. By doing so, Zuckerberg and Chan sidestep the restrictions that govern charitable foundations. Zuckerberg argues that this will give the couple “the flexibility to give to the organizations that will do the best work — regardless of how they're structured”, he wrote in December. No one can fault the pair for wanting to cure disease, but it remains to be seen whether this flexibility will lead to faster cures or just more controversy.

Cardiovascular
University Hospitals - Cleveland
Harrington Investigators